24 May 2023
Amsterdam, May 24, 2023 – In a noteworthy case study published in the Journal of Alzheimer’s Disease investigators report autopsy findings in a 65-year-old woman with Alzheimer’s disease (AD) who received three open label infusions of the experimental anti-amyloid beta (Aβ) antibody drug lecanemab. Four days after the last infusion, she experienced stroke symptoms and died several days later due to multifocal intracerebral hemorrhage despite attempts at therapeutic intervention. Neuropathologic findings reflected therapy-induced Aβ phagocytosis involving fibrillar Aβ both in the parenchymal brain tissue and in the cerebral vasculature.
The most widely explored theory regarding AD pathogenesis is the amyloid cascade hypothesis, which states that Aβ excess in the form of neurotoxins drives the disease process in AD, with neurofibrillary degeneration, neuronal loss, and neurological deterioration occurring as downstream events. By extension, mitigation of Aβ would be a logical strategy for therapeutic intervention.
The patient participated in a phase III study of the efficacy and safety of experimental lecanemab, a humanized monoclonal therapeutic agent thought to target soluble Aβ protofibrils. The trial demonstrated a 27% reduction in the rate of cognitive decline at 18 months. The potential for adverse reactions to Aβ-targeting experimental therapies had been shown in previous clinical trials.
"It is of note that despite clinical trials targeting Aβ have been ongoing for more than 20 years and known adverse reactions clinically and on imaging (amyloid-related imaging abnormalities, or ARIA), we had essentially no insight into cellular reactions to these experimental antibodies or the mechanism of amyloid clearance prior to this case," explained lead investigator Rudolph J. Castellani, MD, Professor of Neuropathology, Department of Pathology, Northwestern University Feinberg School of Medicine, Chicago, IL, USA.
In this case, the patient passed away after only three infusions with lecanemab, in the "subacute” phase of reaction to the drug. This finding at this point in the treatment regimen had not been previously reported. Although autopsy revealed no significant systemic cardiovascular comorbidities, examination of the brain confirmed that the anti-Aβ therapy resulted in a previously undescribed amyloid phagocytic syndrome that extended into the innumerable small blood vessels of the cerebral cortex that had abundant cerebral amyloid angiopathy (CAA). This appeared to have led to the onset of stroke symptoms and precipitated hemorrhage with the attempt at stroke intervention.
Dr. Castellani commented, "In this case it is, in my opinion, abundantly clear that the patient's response to the anti-Aβ therapeutic led to the clinical symptoms and provided a substrate for hemorrhage with therapeutic intervention, raising the issue of a potentially lethal drug interaction.” The question then becomes whether patients receiving anti-Aβ therapeutics can be adequately evaluated for the extent of CAA (which is variable in Alzheimer’s disease, from little to no CAA, to abundant CAA, as in this case), and whether adverse and potentially lethal outcomes can be avoided. On the positive side, there appeared to be partial clearing of Aβ and possibly even phosphorylated tau, the latter not previously described. In short, target engagement was achieved, but it came at the expense of collateral injury to small blood vessels involved by CAA.
Co-investigator Pouya Jamshidi, MD, Department of Pathology, Northwestern University Feinberg School of Medicine, cautioned, “Although this is the first reported case detailing the neuropathologic findings in response to lecanemab, the pattern and distribution of pathology are so striking it is inconceivable to believe this would be an isolated occurrence.”
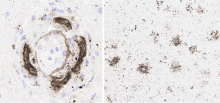
![Caption: The most striking findings were the mononuclear attack on cerebral amyloid angiopathy (histiocytic vasculitis) with fibrinoid necrosis (left, Aβ immunohistochemistry [4G8]), and focally pronounced Aβ plaque phagocytosis (right, CD163 immunohistochemistry), the combination of which does not otherwise exist in nature. Credit: Journal of Alzheimer's Disease.](/sites/default/files/Schermafbeelding%202023-05-24%20123016.png)
Caption: The most striking findings were the mononuclear attack on cerebral amyloid angiopathy (histiocytic vasculitis) with fibrinoid necrosis (left, Aβ immunohistochemistry [4G8]), and focally pronounced Aβ plaque phagocytosis (right, CD163 immunohistochemistry), the combination of which does not otherwise exist in nature. Credit: Journal of Alzheimer's Disease.
According to M.-Marsel Mesulam, MD, Ruth Dunbar Davee Professor of Neuroscience and Chief of Behavioral Neurology, Department of Neurology, Northwestern University, Feinberg School of Medicine, and namesake for the Mesulam Center for Cognitive Neurology and Alzheimer’s Disease at Northwestern, “The availability of lecanemab introduces a new phase in the treatment of AD. The benefits are modest at the group level and unknowable in individual patients. The side effects, even if rarely symptomatic, can be devastating as implied by this case report. Screening for cerebrovascular disease and apolipoprotein E status becomes essential for prescribing the drug. In addition, the patient may need to be told that anticoagulant treatment for stroke, should such an event occur, may become an option with even greater risk.”
"There is clearly a delicate and precarious balancing act going on between Aβ targeting and a potentially deleterious host response, especially as it relates to blood vessels involved by CAA. Better biomarkers that can accurately assess the extent of CAA are badly needed. Neuroimaging and APOE genotyping, while important for risk stratification, leave many cases of severe CAA undetected,” added Dr. Castellani.
“The results of this case call for intense and careful scrutiny of those suffering ill from these experimental drugs to minimize the risk of brain damage and death,” noted George Perry, PhD, Editor-in-Chief, Journal of Alzheimer's Disease, and Semmes Distinguished University Chair in Neurobiology at The University of Texas at San Antonio.
# # #
NOTES FOR EDITORS
The article is “Neuropathology of Anti-Amyloid-β Immunotherapy: A Case Report,” by Rudolph J. Castellani, Elisheva D. Shanes, Matthew McCord, Nicholas J. Reish, Margaret E. Flanagan, M-Marsel Mesulam, and Pouya Jamshidi (https://doi.org/10.3233/JAD-221305), and is published in Volume 93, Issue 2 (May 2023) of the Journal of Alzheimer’s Disease. It is available at https://content.iospress.com/articles/journal-of-alzheimers-disease/jad221305.
To obtain the PDF of the full text or for further information contact Diana Murray, IOS Press, at +1 718-640-5678 or d.murray@iospress.com. Journalists wishing to interview the authors should contact Rudolph J. Castellani, MD, at rudolph.castellani@nm.org.
The examination and case characterization were made possible by permission of the patient's next of kin, and the existing resources of the Department of Pathology, Northwestern Feinberg School of Medicine, and Northwestern Medicine.
ABOUT THE JOURNAL OF ALZHEIMER’S DISEASE (JAD)
The Journal of Alzheimer's Disease (JAD) is an international multidisciplinary journal to facilitate progress in understanding the etiology, pathogenesis, epidemiology, genetics, behavior, treatment, and psychology of Alzheimer's disease. The journal publishes research reports, reviews, short communications, book reviews, and letters-to-the-editor. Groundbreaking research that has appeared in the journal includes novel therapeutic targets, mechanisms of disease, and clinical trial outcomes. JAD has a Journal Impact Factor of 4.106 according to Journal Citation Reports™ (Clarivate, 2022). www.j-alz.com
ABOUT IOS PRESS
IOS Press is an independent international scientific, technical, medical (STM) publishing house established in 1987 in Amsterdam. We produce around 90 journals and 70 books annually in a broad range of subject categories, primarily specializing in health and life sciences (including neurosciences, medical informatics, cancer research, and rehabilitation) and computer sciences (including artificial intelligence, data science, and semantic web). In addition, we offer specialized services that support scientific advancement. www.iospress.com







